Get the product asset
Product Selector
Click-to-answer questions guide you through selecting the right product for your application.

Compliance with complex regulatory requirements – such as FDA 21 CFR Part 11 – requires much more than password protection and restricting user access to files and folders on a corporate network.
ZONE3® Pro has built-in capabilities that directly support the operational requirements of a regulatory-compliant environment. Even if you are not under the jurisdiction of any of the regulatory agencies, these ZONE3 capabilities may ensure adherence to your Quality Management System’s internal requirements for a rigorous metrology environment.
There are nine components of ZONE3 for Regulated Environments –
| User Access Permissions | Locking Projects |
| Audit Trails/Logging | LaunchPad |
| Task Based Programming | Error Handling |
| E-Signatures | Locked UCP Database |
| Expiring Sensor Qualifications |
Additional solutions for qualifying OGP systems in a regulatory-compliant environment:
Equipment Installation Qualification (IQ) Service
The IQ includes analysis and report on verification of documentation, environment, test equipment, artifacts, contact materials and lubricants. Executor identification and deviation reports are provided. The service deliverables include completed documentation forms of equipment installation parameters that can be used towards compliance with FDA IQ requirements.
Equipment Operational Qualification (OQ) Service
The OQ service includes verification of documentation, Operational Verification, Process FMEA, Performance Evaluation, and a Preventive Maintenance Schedule. Executor identification and deviation reports are provided. The service deliverables include completed documentation forms of operational functionality that can be used towards further compliance with FDA OQ requirements.
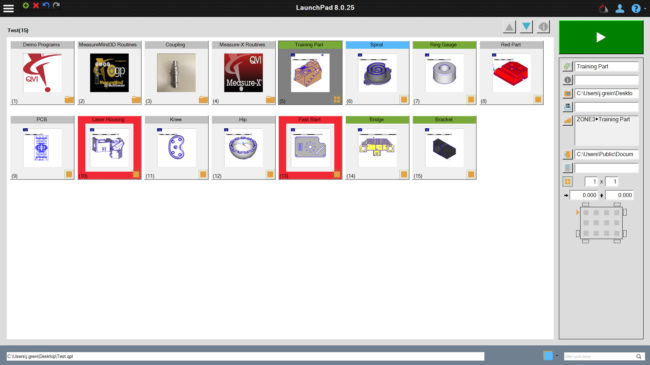 The LaunchPad application is a simplified yet highly configurable user interface where operators with only minimal training can securley launch and run programs.
The LaunchPad application is a simplified yet highly configurable user interface where operators with only minimal training can securley launch and run programs.
Benefits | Features |
|---|---|
| Allow or restrict access to specific functions of ZONE3, or specific software applications outside of ZONE3, based on the individual user | User access permissions – linked to Windows® users – allow for permission sets down to the individual user level |
| Prevent project access, modifications, or editing by unauthorized users | Locking Projects – locks and secures an automated process that is part of a validated production or quality system |
| Easily apply an electronic signature to PDF output of measurement results from the Results Browser, controlled based on user permissions | E-Signatures – in the form of Windows® user credentials or biometric data (e.g., fingerprint) |
| Quickly examine the most recent audit log file, open other log files, search for specific event records and copy from or print log files | Log Viewer™ – an application which allows for viewing audit event records and log files for electronic records. These records consist of audit trail reports generated by ZONE3 and other OGP software |
OGP Authorized Representatives are metrology specialists, qualified to evaluate your measurement requirements and recommend the solution that best fits your needs.
Your local representative is ready to help you with product details, demonstrations, training, or purchase information.
Contact a Sales Rep.





